Avraham O, Deng PY, Jones S, Kuruvilla R, Semenkovich CF, Klyachko VA, Cavalli V. Nature Communications. Sep 29, 2020; 11(1):4891. doi: 10.1038/s41467-020-18642-y.
Abstract
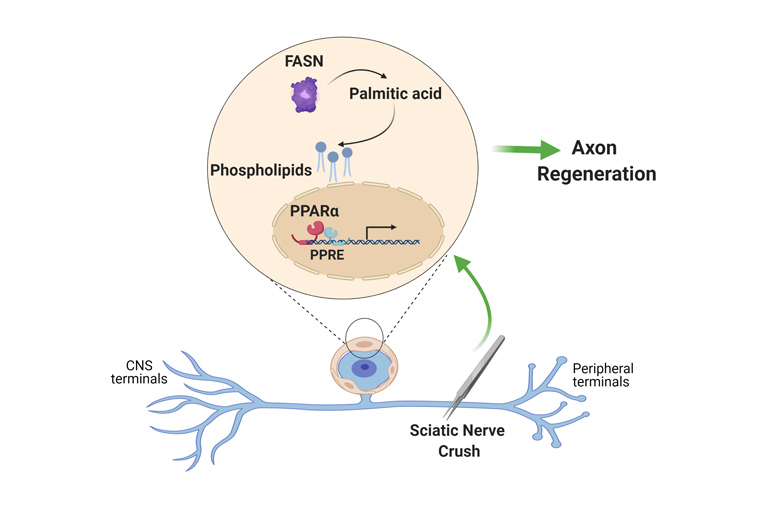
Peripheral sensory neurons regenerate their axon after nerve injury to enable functional recovery. Intrinsic mechanisms operating in sensory neurons are known to regulate nerve repair, but whether satellite glial cells (SGC), which completely envelop the neuronal soma, contribute to nerve regeneration remains unexplored. Using a single cell RNAseq approach, we reveal that SGC are distinct from Schwann cells and share similarities with astrocytes. Nerve injury elicits changes in the expression of genes related to fatty acid synthesis and peroxisome proliferator-activated receptor (PPARα) signaling. Conditional deletion of fatty acid synthase (Fasn) in SGC impairs axon regeneration. The PPARα agonist fenofibrate rescues the impaired axon regeneration in mice lacking Fasn in SGC. These results indicate that PPARα activity downstream of FASN in SGC contributes to promote axon regeneration in adult peripheral nerves and highlight that the sensory neuron and its surrounding glial coat form a functional unit that orchestrates nerve repair.
Department mini interview
In this article, you describe the key role of a relatively poorly studied type of glial cells, satellite glial cells (SGC), in nerve repair. Does this contradict the prevailing view that axonal repair is a neuron-intrinsic mechanism?
Valeria Cavalli: No, it does not contradict the notion of neuron intrinsic mechanisms. I think it expands the notion that some of the neuronal response to injury might be guided by the surrounding environment, especially these satellite glial cells that completely enwrap the neuronal soma.
What prompted this particular study? How did it start?
Oshri Avraham: Dorsal root ganglia have been studied for decades to understand the neuronal responses to nerve injury and identify mechanisms that can promote neural repair. Because sensory neurons are the largest cells in that tissue, it has been assumed that any changes observed in the ganglia are due to changes occurring in neurons. We decided to examine this in more detail and focused on satellite glial cells, because they are so closely associated with neurons. We hypothesized that these cells might also respond to nerve injury and potentially contribute to the nerve repair process.
Can you tell us a bit more about these intriguing satellite glial cells? Are they a class of glial cells on their own? Do they solely exist in DRGs?
Oshri Avraham: Yes, satellite glial cells represent a glial cell population on its own. Satellite glial cells exist in other ganglia, such as the spiral ganglion in the ear, the trigeminal ganglia innervating the face and neck or the sympathetic ganglia. There are two main types of glial cells in the peripheral nervous system: Schwann cells and satellite glial cells. They have the same origin and develop from neural crest cells, but have different morphologies and molecular signatures. Schwann cells wrap the axon, whereas satellite glial cells surround the neuronal soma. Satellite glial cells are well known in the field of pain, as they contribute to control nociceptive thresholds. Some of their function are similar to astrocytes in the central nervous system, so we could call them peripheral astrocytes.
They occupy such a unique position, what do you think is their function?
Valeria Cavalli: Yes, their morphology is unique. Multiple satellite glial cells completely surround the neuronal soma. This unique morphology can inform us on their function. Since sensory neurons reside outside the blood brain barrier, I believe that satellite glial cells could represent a form of barrier, in addition to their function in regulating neuronal excitability and providing trophic support to the neurons.
How do your results advance the understanding of nerve regeneration? Do you believe that the interplay between sensory neurons and satellite glia holds the key to elucidating other constrains surrounding axonal repair?
Valeria Cavalli: I think that nerve repair is a complex process that requires interaction between the neuron and its environment. We just got new data that satellite glial cells do not have the same response if we injure the central sensory axon, suggesting that these cells could hold part of the key to neural repair. Combinatorial therapies targeting the neuron and its environment, including satellite glial cells, could make a difference for neural repair.